History
Discovering the structure of the atom was a monumental
feat, one that spanned several centuries and involved many of the greatest minds
of our times. Since Mendeleev's creation of the periodic table in 1869,
scientists were mesmerized with the periodic variation that appeared in the
table and strived to create a model that would explain the observed phenomenon.
It was not until the 1920's that the answer was finally discovered.
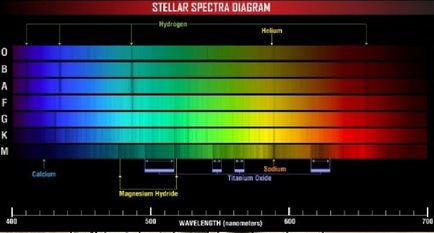
Scientists had observed, as early as the 1800's, that if metal was exposed to light at specific frequencies, electrons would be ejected from the metal. This property of matter bewildered chemists for many years and it was only in 1905 that a brilliant, young physicist by the name of Albert Einstein managed to provide the answer. Einstein noted that when atoms were energetically excited they would give off light, but unlike the white light from the sun, the light emitted by atoms had discrete wavelengths.
Instead of a rainbow of colours, an atom can only emit a certain number of distinct colours. These colours, when placed on a dark background, form a line spectrum and each element has its own, unique spectrum. Sodium, for example, will give off several shades of yellow light while hydrogen can give off indigo and red light. The atoms would only give off light when struck by light with a specific frequency, leading Einstein to conclude that the atom's electrons and the packets of light striking them somehow interacted. As well, the distinct bands of colour that were expelled indicated that the energy emitted by the atom was constrained to only a few specific levels.
Instead of a rainbow of colours, an atom can only emit a certain number of distinct colours. These colours, when placed on a dark background, form a line spectrum and each element has its own, unique spectrum. Sodium, for example, will give off several shades of yellow light while hydrogen can give off indigo and red light. The atoms would only give off light when struck by light with a specific frequency, leading Einstein to conclude that the atom's electrons and the packets of light striking them somehow interacted. As well, the distinct bands of colour that were expelled indicated that the energy emitted by the atom was constrained to only a few specific levels.
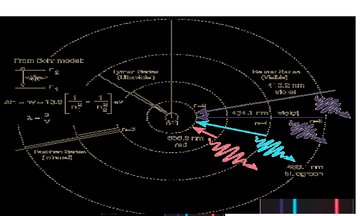
Inspired by this insight, Einstein’s friend and colleague, Niels Bohr, proposed the now famous model of the hydrogen atom, describing it as a nucleus with an
electron circling around. According to Bohr, an electron required a specific
amount of energy to continue spinning in an orbital and the observed emission
of spectral lines was the energy released as a result of the electron falling from
a higher-energy orbital to a lower energy orbital. Since the difference in energy
levels between orbital’s is distinct, the specific bands of light observed is explained by this model. Though the Bohr model was instrumental in eventually discovering the structure of the atom, it failed to accurately predict the behaviour of elements past Hydrogen and is remembered today, not for being correct, but for being the first model that recognized that electrons occupied specific energy levels and didn't roam freely around the nucleus.
electron circling around. According to Bohr, an electron required a specific
amount of energy to continue spinning in an orbital and the observed emission
of spectral lines was the energy released as a result of the electron falling from
a higher-energy orbital to a lower energy orbital. Since the difference in energy
levels between orbital’s is distinct, the specific bands of light observed is explained by this model. Though the Bohr model was instrumental in eventually discovering the structure of the atom, it failed to accurately predict the behaviour of elements past Hydrogen and is remembered today, not for being correct, but for being the first model that recognized that electrons occupied specific energy levels and didn't roam freely around the nucleus.
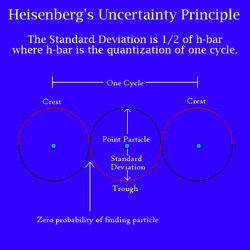
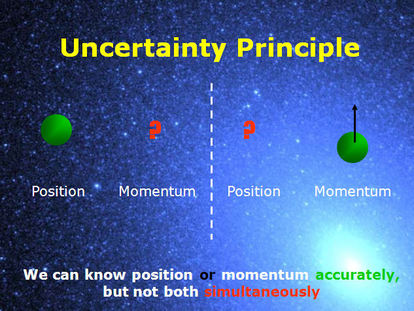
Around the same time Werner Heisenberg proved that it was impossible to know the precise location of an electron as well as its momentum, a statement known as Heisenberg’s Uncertainty Principle. This principle can be understood by considering what would occur if we tried to find an electron’s location at a moment of time. To “see” the electron we would need to bounce light off the particle, but as Einstein proposed, the interaction with light’s photons would transfer energy onto the electron. The increase in energy would make the electron move faster and by the time the light returned to our eye, the particle would have a different momentum and position then the one observed.
This statement about matter is represented by the mathematical equation:
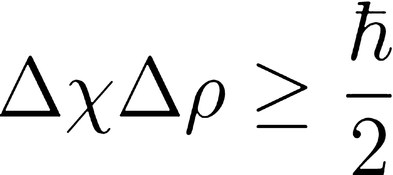
where...
h is Planck's constant reflecting the size of energy
quanta approximately equal to 6.626*10^(-34) kg*m^2/s.
Δx is the standard deviation of position
Δp is the standard deviation of momentum
|
From the equation it is evident that as our certainty of momentum (∆mv) increases, so too does the uncertainty of position(∆x) , and vice versa. Hence we can never know both the position and momentum of any object beyond a certain degree of precision and the very act of determining these values will cause them to change. |
Erwin Schrodinger, realizing that all attempts to model the motion of an electron as a particle would fail, made the final breakthrough when he instead decided to look
at the electron as a wave. He designed an equation modelling the electron
and called its solutions orbitals, represented by the symbol ψ.
These solutions would tell where the electron would most probably
be and the chance of finding the electron within a given volume around
the nucleus. When the solutions are graphed, we gain a sense of how the atom
truly looks like. Schrodinger provided the final piece of the puzzle and laid
the problem of describing the structure of the atom to rest.