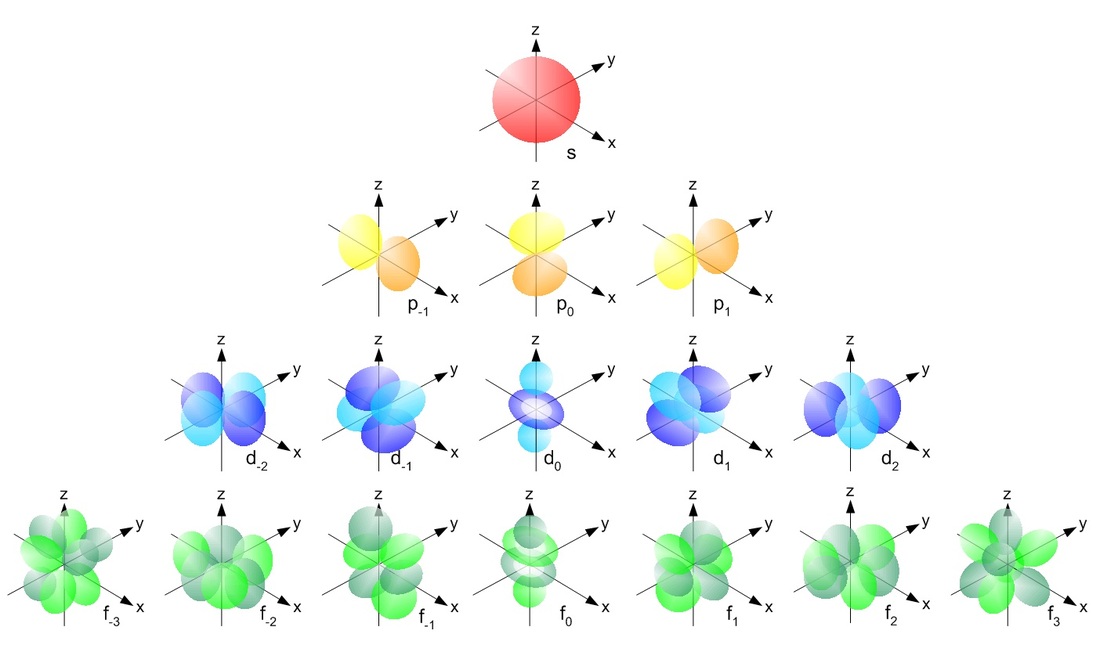
s orbitals orbitals are spherical and have only 1 possible
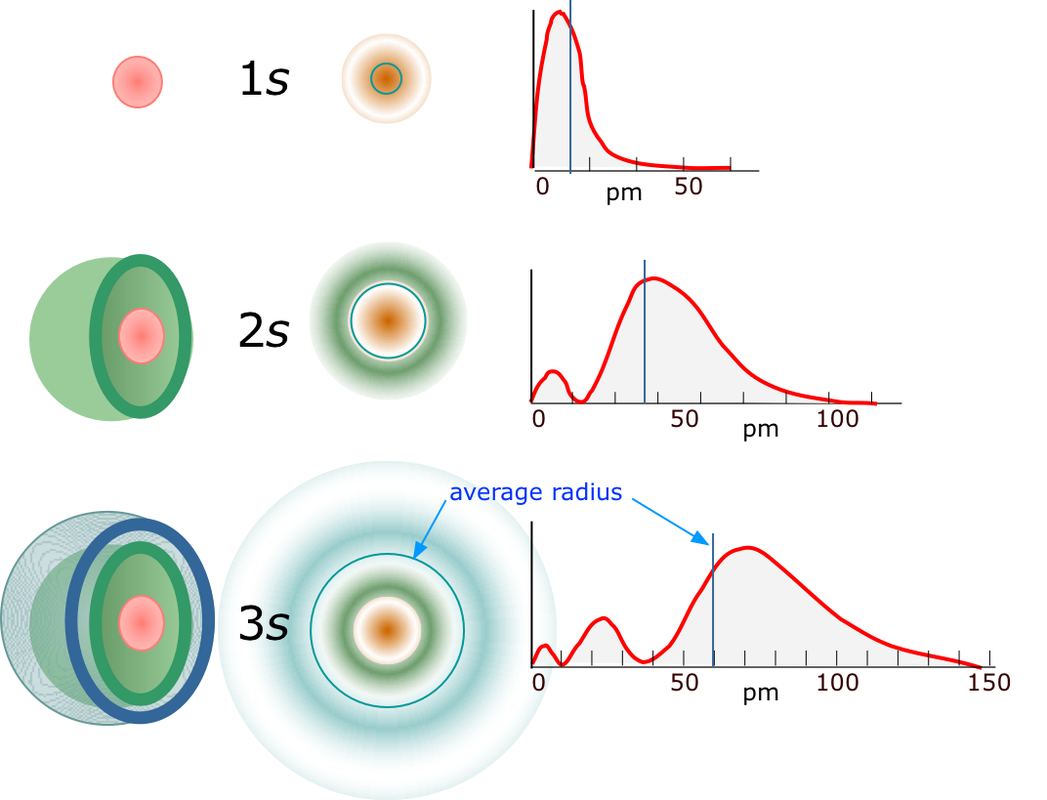
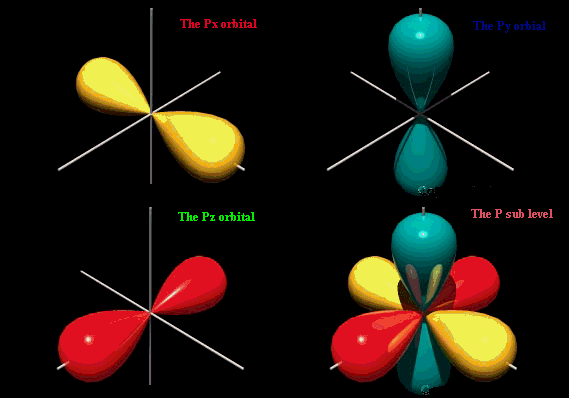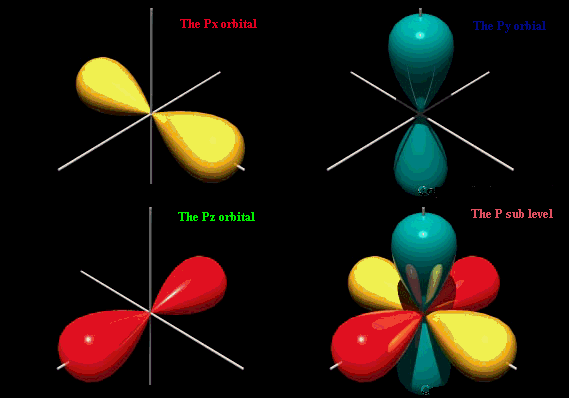
orientation implying that they can only have ml=0. There is only 1 s orbital per shell. The probability of finding the electron is greatest around the nucleus and decreases rapidly as you move away from it, though it never does reach zero. As a result, no atom has a defined size and the drawn shape merely indicates where the electron will spend the majority, but not all, of its time. s orbitals in higher shells have a greater size and the electrons occupying these orbitals must therefore be a greater distance from the nucleus compared to the electrons in smaller s orbitals. The s orbital will hold a total of 2 electrons. p orbitalsp orbitals are dumbbell shaped compared to the spherical shape of its predecessor with the electron being equally likely to occupy lobes on either sides of the nucleus. Due to its shape, the probability of finding the electron at the nucleus is zero. There are 3 allowable ml values since l=1 and the p orbital is first present in the second shell. The orbitals rest along the x, y and z axis depending on the value of ml. The 3 p orbitals will hold a total of 6 electrons.
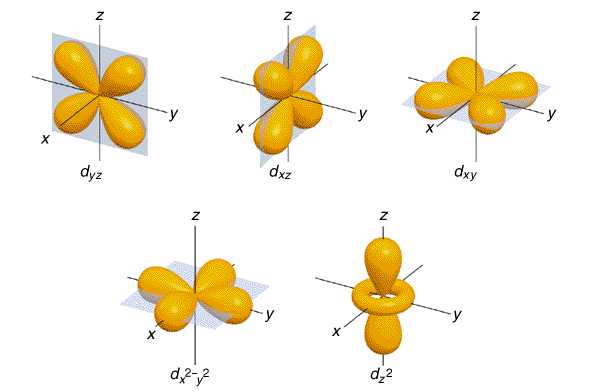
d and f orbitalsThe third and higher shells have d orbitals which distinguish themselves from the previous orbitals by having two different shapes. Four of the five orbitals are shaped like cloverleafs, each having four areas of maximum electron probability. Like the p orbital, the nucleus is a point with zero electron probability. The fifth d orbital differs from all the others and shares more similarities with the pz orbital but has an additional donut shaped region around the origin. Despite their varying shape, the electrons within all 5 d orbitals have the same energy levels. The 5 d orbitals will hold a total of 10 electrons
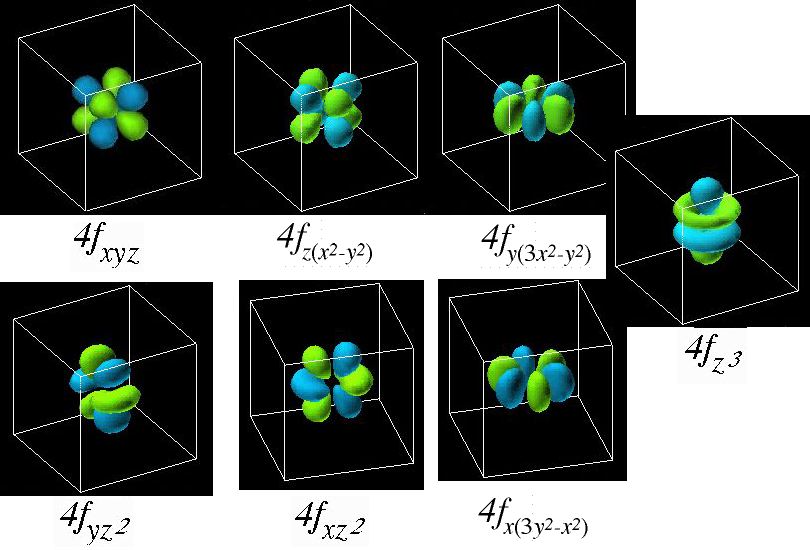
The f orbitals are found in shells with a quantum number of 4 or higher and there shape is more complex then all the rest boasting eight regions of maximum electron probability, again with the nucleus being the site where the electron will never be found. The 8 f orbitals will hold a total of 16 electrons. The diagram below summarizes the shapes of the s, p, d and f orbitals
|