The Quantum Number
A wave function can be described with four variables, called
quantum numbers, denoted n, l, ml and ms. Together they
describe the energy level and the three-dimensional shape of the
area the electron occupies.
quantum numbers, denoted n, l, ml and ms. Together they
describe the energy level and the three-dimensional shape of the
area the electron occupies.
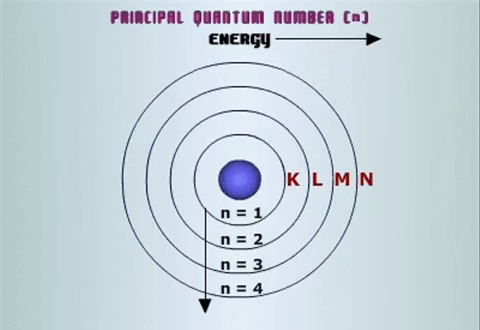
The Principal Quantum Number, nThe Principle Quantum Number (n) is a positive integer which determines the size and energy level of the orbital. As n increases, the number of orbitals in a given shell as well as their size gets larger. Since energy is required to separate the negatively charged electron and positively charged nucleus, the further the electron from the atom’s center, the more energy it must have and so the electrons occupying orbitals with higher values of n have higher energy levels. For atoms with a single electron, like Hydrogen or He+, the energy of an orbital depends solely on the Principle Quantum Number. For other atoms, quantum number l plays a part in determining the energy of the orbital. We will see why shortly. The quantum number n is often used to label shells in the atom and so orbitals with quantum number n are said to occupy the nth shell.
|
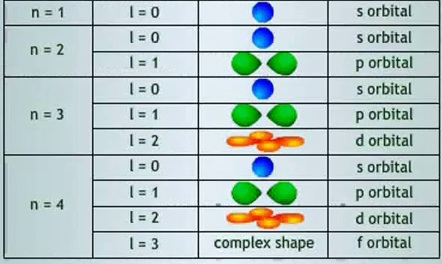
The Secondary Quantum Number, lThe Angular-Momentum Quantum Number (l) describes the three dimensional shape of an orbital. An orbital with a Principle Quantum number n can have an angular momentum quantum number l from the
integers 0 to n-1. Within each shell, there are n different orbitals, sometimes called subshells, each with a distinct shape. For instance: in the n=1 shell, there is an l=0 orbital and within an n=2 shell, there is an l=0 and l=1 orbital. Each number of l refers to a specific subshell shape and usually the number 0, 1, 2, 3… are replaced with the letters s, p, d, f… respectively. After f, subshells are named alphabetically, starting with g, h etc. For example, we would call an orbital with n=2 and l=1 a 2p orbital. 2 designates the shell and p describes the subshell shape |
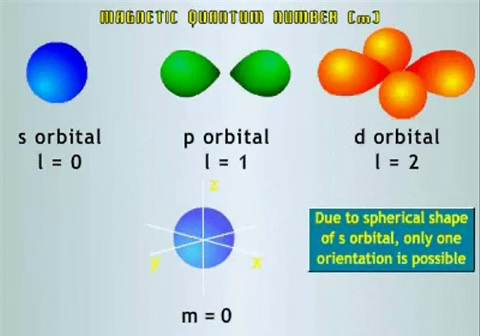
The Magnetic Quantum Number, mlThe Magnetic Quantum Number (ml) describes the configuration of the orbitals according to a set of axes in three dimensions. For any l, the magnetic quantum number can take on integer values from –l to +l. Hence there are 2l+1 different orientations for each subshell. For example, if l=0, ml=0 and if l=1, ml can equal -1, 0, or +1.
|
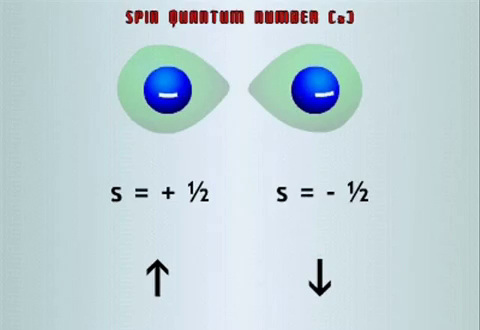
The Spin Quantum Number, msThe Spin Quantum number (ms) is the fourth quantum number and describes the direction the electron spins. Electrons can
be envisioned, like our planet, as spinning around an axis. However unlike Earth, electrons can spin in either the clockwise or counter clockwise direction. The spinning charge will create a magnetic field, which though tiny, has profound effects on the sub atomic behaviour of the atom. The spin quantum number can either have the value +1/2 or -1/2. +1/2 ndicates that the electron is spinning counter clockwise while -1/2 indicates the electron is spinning clockwise. +1/2 is usually represented as an upward arrow while -1/2 is represented with a downward arrow. The importance of this quantum number becomes evident when we consider Pauli’s exclusion principle which states the no electron in an atom can bear the same four quantum numbers. This principle is very important when trying to identify the orbitals filled by electrons. Electrons occupying the same orbital have the same n, l and ml values but have different ms values, either +1/2 or -1/2. We can deduce from this principle that each orbital can house no more then 2 electrons, each with an opposite spin. |
The table below summarizes the above information and show
the combinations of quantum numbers for the first four shells.
the combinations of quantum numbers for the first four shells.
Combinations of Quantum Numbers for the first Four Shells